Colonization by respiratory pathogens

Dynamics of nasopharyngeal colonization by potential respiratory pathogens
Studies have shown that colonization of the nasopharynx by potential respiratory pathogens Streptococcus pneumoniae, Haemophilus influenzae and Moraxella catarrhalis is established early in childhood, although rates vary greatly according to locality, sampling frequency, individual and social factors. Factors influencing colonization and elimination are not as yet fully understood, but adhesion to mucosal receptors and immune responses are implicated in addition to bacterial properties and colonization resistance dynamics.
Colonization in children and adults has been intensively studied in various localities. Potential pathogens are more likelyto colonize the nasopharynx of children prone to recurrent otitis media, where impaired local immunity and repeated exposure to respiratory pathogens are additional risk factors. Adults with chronic respiratory tract disease also have higher carriage rates. The factors contributing
to increased risk of carriage of potential respiratory pathogens, as well as to clinical infection and antimicrobial resistance, are summarized in this review.
Introduction
The normal fetus is sterile as long as its membranes remain intact. Immediately after birth, the neonate rapidly acquires commensal bacteria that colonize skin and mucosal sites. The normal upper airway rapidly becomes colonized and is a major ecological reservoir of bacterial species.1 This bacterial flora is in a constant state of flux: bacteria are acquired, eliminated and re-acquired many times over during life.2–7 In spite of this, and although the details of the flora may differ between individuals (e.g. subtype, proportions, age of acquisi- tion), the general patterns are the same.8
In children the nasopharyngeal flora becomes established during the first year of life.3,9,10 The nasopharynx is densely colonized by a broad variety of microorganisms, including commensal bacteria as well as potential pathogens such as Streptococcus pneumoniae, Haemophilus influenzae (essen- tially non-typeable strains) and Moraxella catarrhalis. Probably all humans are colonized with these potential respira- tory pathogens at least once early in life. In most cases these organisms are carried without causing clinical symptoms. However, when the condition of the host is altered, micro- organisms may invade adjacent sites and/or invade the bloodstream, causing disease. Nasopharyngeal colonization
precedes invasion but identifiable disease occurs in only a small percentage of persons who are colonized. On the other hand, the nasopharynx is a major source of secretions contain- ing bacteria that spread between individuals and may become pathogens. In this respect, nasopharyngeal carriage is import- ant, since it is related to both development of disease and spread of the pathogens.
- S. pneumoniae and M. catarrhalis colonize as many as 54% and 72% of children, respectively, by 1 year of age.10 Routine vaccination of young children with influenzae type b has reduced the prevalence of nasopharyngeal colonization with this respiratory pathogen.11–14 Colonization with non- typeable H. influenzae in the first 2 years of life continues to be high, as demonstrated by the work of Faden et al.,3,9 who showed that 44% of children become colonized by the age of 2 years, with the most rapid acquisition rate occurring in the first year (34%).
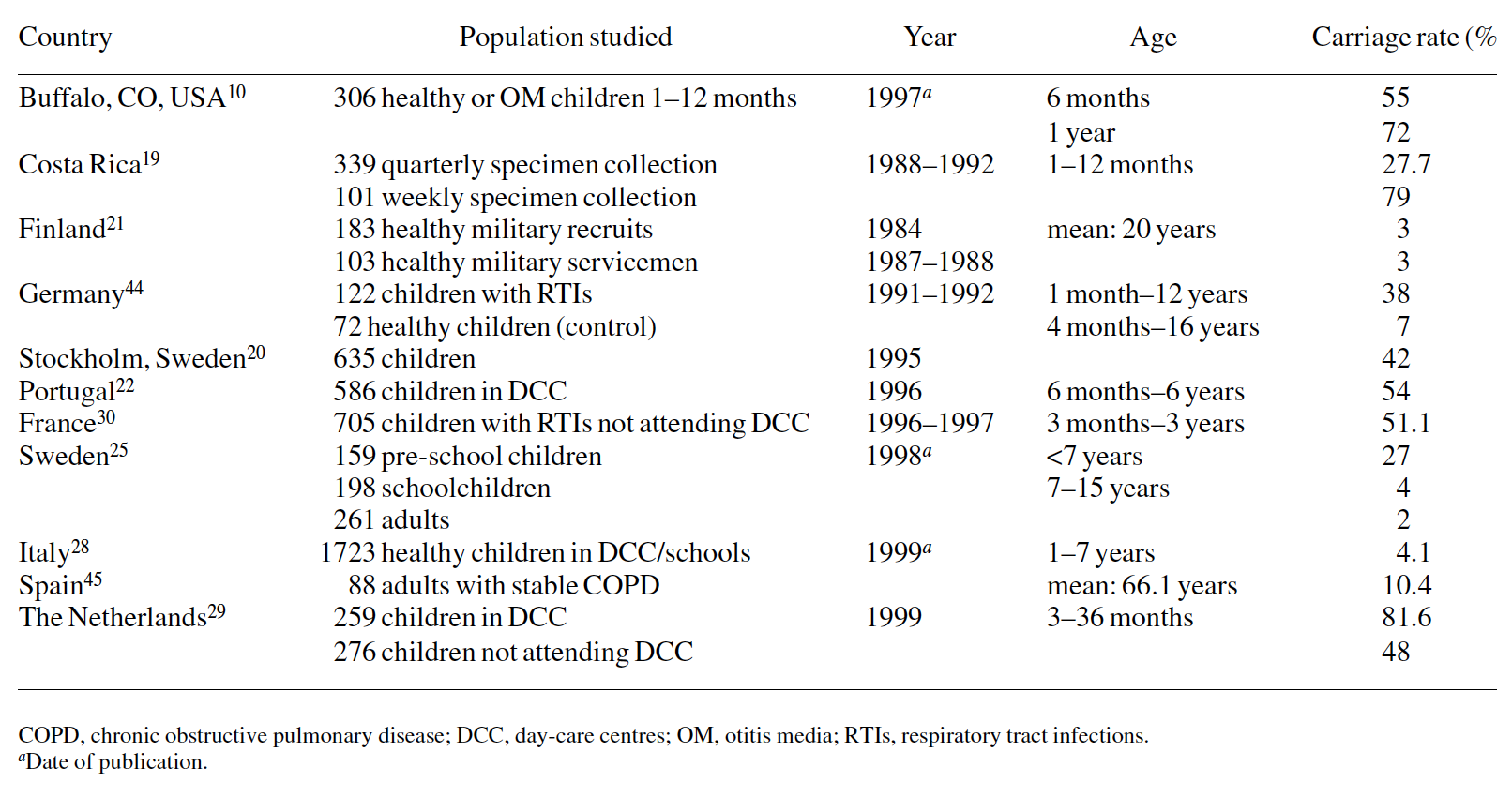
The reported rates of bacterial acquisition and carriage vary extensively between different studies and geographical sites,3,10,13,15–45 (Tables 1–3). These differences have been related to genetic background variables and socio-economic conditions including housing, access to health care, poor hygiene, family size, overcrowded living conditions, day-care contact, number of siblings, etc.
Table 1. Nasopharyngeal carriage of S. pneumoniae
Table 3. Nasopharyngeal carriage of M. catarrhalis
The relative role of social factors versus individual variation in resistance to colonization is not clear, although the results from several studies have suggested that both acquisition and carriage status in newborns and infants are influenced in great measure by the socio-economic status of the study population. Almost all Australian Aboriginal children are colonized by M. catarrhalis and/or pneumococci and/or H. influenzae by 1 month of age.46 In contrast, non-Aboriginal children are less often colonized (H. influenzae, 1.5%; M. catarrhalis, 6.1%). Only 50% of children from Stockholm and 43% of Finnish children are colonized by S. pneumoniae at 2 years of age, the group with the highest prevalence of carriage.20,47 Aboriginal infants, however, are colonized by M. catarrhalis as early as 8 days of age and by H. influenzae and S. pneumoniae as early as 10 and 20 days, respectively. Non-Aboriginal children are colonized by M. catarrhalis, H. influenzae and S. pneumoniae at a median age of 200, 209 and 270 days, respectively. In addition, Aboriginal children are typically colonized with multiple species of pathogenic respiratory bacteria.46
However, even in countries with comparable environments the rates of colonization vary widely in different reports as demonstrated in two studies carried out in Italy and Portugal in healthy pre-school children; 3.5% versus 47% of all infants in these countries, respectively, were colonized with S. pneumoniae, 11.9% compared with 72% with H. influenzae, and 4.1% compared with 54% with M. catarrhalis, respectively.22,28 It is even possible to find marked differences in bacterial carriage and resistance patterns between day-care centres located within a small area.35
These differences suggest that the factors that influence colonization are multiple and not entirely clear. There is no doubt that the local host immune response plays an important regulatory role in the trafficking of pathogens in the upper airway.3 Otitis-prone children generate a poor local immune response to non-typeable H. influenzae, which leads to persistent and recurrent colonization. In contrast, children who become colonized briefly tend to generate a good local immune response.48 Serotypes of S. pneumoniae that are poorly immunogenic tend to colonize for longer periods.2 Also, the absence of transmission of pneumococci from children to their parents and other adults in a closed community in Israel and the low carriage rates in adults suggest the existence of immunological protection in the latter.19–21,25,34
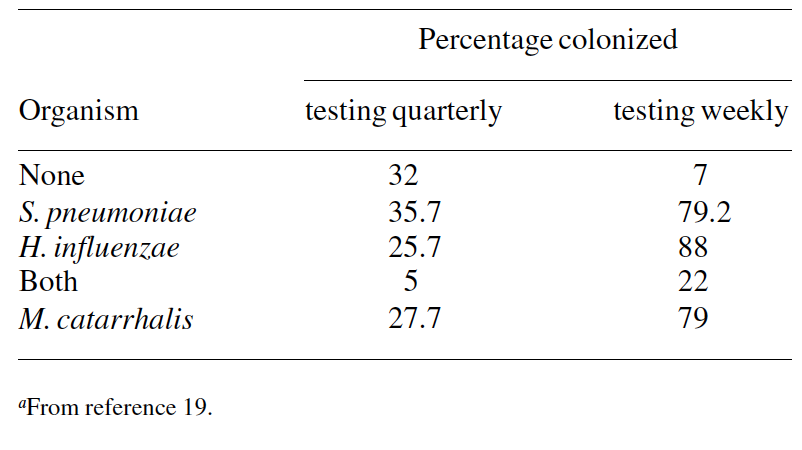
Methodological factors (quality of the sampling, culture techniques, number and frequency of specimen collection) can also be responsible for the observed differences in reported colonization rates.49 On a single occasion, cultures yielded pneumococci in 5–10% of healthy adults and 20–40% of healthy children. With repeated culture the isolation rate increased in all age groups, rising to 40–60% in children.50
Data from Costa Rica illustrate this concern and show the existence of marked differences in colonization rates related to the frequency of specimen collection (Table 4).19
Bacterium–host interactions
Potential respiratory pathogens gain entry into the host by colonizing the nasopharyngeal mucosal epithelium. Adhesion is the initial event in the colonization process and implies the attachment of bacterial surface proteins to host cell carbo- hydrate receptors.51
The attachment of S. pneumoniae is mediated through specific bacterial surface adhesin molecules (such as pneumo- coccal surface antigen A, pneumococcal surface adhesin A and choline-binding proteins) that form a bridge between bacterial surface components and epithelial cell receptors. Multiple ligand–receptor pairings occur at different levels of the respiratory tract, but there are site-specific differences in the relative importance of these pairings. The exact structure of the pneumococcal adhesins responsible for binding is not entirely known but the N-acetylgalactosamine β1-4 galactose (GalNAcβ1-4Gal) moiety of glycolipids has been identified as a prominent epithelial cell receptor for pneumococcal attachment.52 Sialic acid is a prominent ligand in the upper respiratory tract.53 Fibronectin and other matrix proteins may also serve as targets for pneumococcal binding.54 Phase vari- ation may also play a role. While the transparent variant is key to colonization, the opaque form exhibits improved survival in the blood. The mechanism for the different adhesive and invasive capabilities between opaque and transparent forms involves modification of the cell wall structure.55,56
Transparent pneumococcal variants have considerably more phosphorylcholene, which contributes to their capacity to adhere to mammalian cells perhaps via the receptor for platelet- activating factor.57,58 On the other hand, transparent variants
bind to N-acetylglucosamine β1-3 galactose between 102 and
106 times more efficiently than their opaque counterparts.59
- influenzae determinants of adhesion include fimbriae, outer membrane proteins (OMPs, such as OMP P5, OMP P2, HMW1 and HMW2 adhesins, Hia, Hap, Hsf, OapA, PE binding adhesin) and lipopolysaccharide, each of which has its own specificity for host molecules. P2 and P5 OMPs of H. influenzae adhere to mucin.60 Fimbriae bind to carbohydrate residues of glycoproteins or glycolipids.61 Also, afimbriated strains of non-typeable strains may adhere to oropharyngeal epithelial cells, suggesting the presence of non-fimbrial adhesins. High-molecular-weight surface-exposed proteins (HMW1 and HMW2) interact with distinct eukaryotic cell receptors and perhaps function at different steps in coloniza- tion. HMW1 recognizes a glycoprotein receptor containing N-linked oligosaccharide chains with sialic acid.62 HMW2 interacts with an uncharacterized carbohydrate moiety.61
Little is known about the adherence of M. catarrhalis. The outer membrane protein UspA1 is essential for the attachment of M. catarrhalis to host epithelial cells according to data from several studies.63,64 This high-molecular-weight OMP is capable of binding to the extracellular matrix proteins fibronectin and vitronectin.63 It has also been shown that mutants defective in expression of UspA1 exhibit decreased adherence to several cell types.64 Haemagglutinin OMP 106 may play a role in adherence and is able to bind to the glycolipid globotetraosylceramide.65 CD protein, a homologue of Pseudomonas OprF protein, binds specifically to human nasal and middle ear mucins but not to salivary mucin.65
Table 4. Nasopharyngeal colonization during the first year of life a
Immune response
Typically the pathogens remain in the airway for several months before disappearing or being replaced by a different strain of the same pathogen. Although the total host factors responsible for elimination of the pathogens are not well understood, recent data suggest that a brisk local immune response to an organism would prevent colonization and limit its duration, whereas a poor immune response would result in more prolonged carriage.3,66 In general terms, mucosal immunity matures earlier than systemic immunity and the magnitude of response affects the colonization pattern.2
On the other hand, many different strains of each pathogen may colonize the host. A good immune response to a highly conserved surface antigen (PspA of S. pneumoniae, P4 and P6 of H. influenzae, UspA of M. catarrhalis) would result in local protection against a number of different strains, while a good immune response to a strain-specific surface antigen (P2 of H. influenzae) would prevent recolonization with homo- logous but not heterologous strains.66
Nasopharyngeal colonization stimulates the production of local IgA and systemic IgG responses to pneumococcal surface protein A (PspA), pneumococcal surface adhesin A (PsaA) and to pneumolysin (Ply).67,68
A higher proportion of IgA-positive nasopharyngeal samples and higher antibody concentrations are observed in children with pneumococci in nasopharyngeal samples or middle ear fluid than in children with cultures negative for pneumococci. Also, detection of antibody is possible as early as 6 months of age.67 These results differ from those observed by Samukawa et al.,69 who found that a low percentage of children colonized with S. pneumoniae had detectable specific IgA to PspA at the time of colonization as well as from 1 to 5 months after colonization
The serum antibody response to PspA, PsaA and Ply may be detected in children at 6 months of age. However, IgG PspA remains at relatively low levels until the age of 2 years.69 The antibody concentrations increase with age and are strongly associated with pneumococcal exposure, whether by carriage or infection (acute otitis media, AOM).68,70 However, adult levels drop to those observed at 3 years of age. This may explain the heightened susceptibility of certain adults to pneumococcal infection.66
Children also develop an immune response to H. influen- zae that influences colonization as well as infection. Prior to colonization, 0% of children possess mucosal antibody (IgA) to H. influenzae; this rises to 100% after colonization.48 The rapid disappearance of the initial colonizing strain of H. influ- enzae shown by Faden et al.3 suggests an early secretory anti- body response that eliminates the pathogen and prevents the re-acquisition of this strain. During colonization and there- after the proportion of children exhibiting strain-specific serum bactericidal antibody rises progressively to 100%.66
The serum immune response to surface protein UspA of M. catarrhalis has been studied in several age groups. Levels of IgG UspA remain relatively low during the first 2 years of life.69 Thereafter they rise gradually and peak during adult- hood.69 IgA UspA-specific antibody is not detected in the respiratory tracts of children colonized in the first 6 months of life.69
Effect of commensal nasopharyngeal microorganisms:
bacterial antagonism
Resident nasopharyngeal viridans streptococci can antagon- ize colonization by other streptococci. This concern has been best described between Group A β-haemolytic streptococci and viridans streptococci, but inhibition of S. pneumoniae,
- influenzae and M. catarrhalis growth by α-haemolytic
streptococci has also been demonstrated in vitro in healthy children, children with secretory otitis media and children with recurrent otitis media. In healthy children, α-haemolytic streptococci are able to inhibit 92% of the S. pneumoniae isolates, 74% of the non-typeable H. influenzae isolates and 89% of the M. catarrhalis isolates. In children with both secretory and recurrent otitis media, α-haemolytic streptococci are significantly less capable of inhibiting S. pneumo- niae (73%) and H. influenzae (58% and 54%, respectively). However, there is no difference in the inhibitory activity of α-haemolytic streptococci against M. catarrhalis.71
These data support the importance of bacterial antagonism in maintaining the balance between components of the resident flora and the transient invaders.
Epidemiology
The mean age of first acquisition of S. pneumoniae and/or H. influenzae and/or M. catarrhalis is 6 months with a range of 1–30 months,2,10,17,31,38,43,46,72 although in particularly predisposed populations it can be as early as 8–10 days.46
At 1 year of age, 50–100% of infants are colonized with a respiratory pathogen.10,19,31,33,34,39,43,46 Simultaneous colon- ization with multiple species of potentially pathogenic respiratory bacteria is frequent,73 mainly in predisposed populations.31,43,44,46
Although variable, the colonization rates stay high during childhood and diminish in adults. Usually, bacteria are intro- duced by children into a household and spread to other family members. However, it has been shown that carriage is signifi- cantly lower in mothers than in their children.19 Also, a recent study carried out in a kibbutz in Israel indicated that pneumo- cocci do not appear to be transmitted from children to their parents or to other adults, despite the high degree of interper- sonal contact occurring in this closed community. This sug- gests the existence of an effective immunological barrier. In addition, socio-economic factors seem unlikely to influence colonization rates in a kibbutz community where living stand- ards are comparable.34
Essentially three patterns of carriage of each species can be distinguished: rapid elimination of the initial strain, prolonged colonization with the initial strain and sequential colonization with different strains.3 Simultaneous carriage of more than one genetically dis- tinct isolate of S. pneumoniae, H. influenzae or M. catarrhalis has been observed. This may result from inadequate clearance of colonizing bacteria or from exposure to multiple circulat- ing strains.46 Dhooge et al.74 have shown a close association between risk factors for higher colonization and carriage with multiple genetic types of bacteria. More than 90 serotypes of S. pneumoniae have been identified on the basis of antigenic differences in their capsular polysaccharides. Some children are never colonized with S. pneumoniae while others became colonized with up to four different types during the first year of life.31,38,66,75 The most common serotypes found in children are 6, 14, 19 and 23.15,29,30,36,39,47,76,77 Fifty per cent of children who become colonized by H. influenzae in the first year become colonized with only one strain while others become colonized sequentially with as many as seven different strains.3,38,43,75,78 Adults typically carry only one strain, while children often harbour multiple distinct strains concur- rently.3,61 A similar pattern is observed with M. catarrhalis since in the first 2 years of life a child acquires and clears three or four different strains of this organism.4
The duration of colonization with non-typeable H. influen- zae may vary from 2 weeks to 5 months.3,43,74 Within 3 months 90% of the initial strains became undetectable and 78% of children were colonized for <2 months.3 Spinola et al.79 analysed nasopharyngeal isolates of non-typeable H. influenzae collected longitudinally from three children in a day-care setting and observed that the children who carried a strain for several months subsequently lost it, and acquired a new strain; re-acquisition of a strain carried previously was uncommon. Colonization with pneumococci can be detected for ∼1–4 months.31,38,80 Serotypes that induce weak immunity, such as 6 and 23, tend to colonize for longer periods.2 The clearance of colonization in disadvantaged populations such as Aboriginal children is impaired because of overcrowded living conditions, high carriage rates and multiple circulating bacterial strains.46 The duration of carriage is inversely cor- related with age. In adults an individual serotype usually persists for short periods, ∼2–4 weeks, but sometimes much longer.80
More investigation is needed to explain whether the turn- over of species is due to colonization of the nasopharynx by new types or unmasking of organisms that were already present but were not detected because of heavy colonization by other types.29,75
Factors affecting colonization
Many factors influence nasopharyngeal carriage rates: age, season, type of child day care, number of siblings, acute respiratory illness, diet (breast-feeding versus bottle-feeding), sleeping position. In adults, other factors are implicated: presence of children at home or at the workplace, chronic obstructive pulmonary disease (COPD), obesity, immunosuppression, allergic conditions, acute sinusitis, etc.
Age. In general, the prevalence of carriage of pathogenic bacteria in healthy individuals decreases with age. These bac- teria are often present in nasopharyngeal samples taken from healthy pre-school children (<7 years old), but rarely from people of >16 years of age.22,25 The overall isolation frequencies for pre-school children, schoolchildren and adults found by Gunnarsson et al.25 in Sweden are: 27%, 4% and 2% for M. catarrhalis, 19%, 6% and 0. 8% for S. pneumoniae and 13%, 6% and 3% for H. influenzae.
Faden et al.10 evaluated nasopharyngeal colonization during the first year of life. This study demonstrated that at 6 months of age, 26% of infants are colonized with M. catarrhalis, 24% with S. pneumoniae and 9% with H. influenzae. At 1 year of age these percentages increased to 72%, 54% and 33%, respectively. In another study,4 the same authors also showed that 66% of infants became colonized with M. catar- rhalis within the first year of life, increasing to 77.5% by 2 years of age. This fact is of great importance, since there is a direct relationship between the frequency of colonization by potential respiratory pathogens and the frequency of acute otitis media (AOM). On the other hand, early colonization is associated with early first episodes of AOM.10 In general, colonization rises gradually and peaks at 2–3 years of life, then decreases gradually until 15–16 years of life, eventually increasing again in adults older than 65 years.21,22,45,47,81,82
Although variable, the colonization rates in healthy adults are lower than in children (Tables 1–3). However, some stud- ies have shown higher rates of colonization. S. pneumoniae colonizes the nasopharynx of up to 40% of healthy adults and carriage of up to four different serotypes for several months has been documented.51 Other reported carriage rates in various age groups are shown in Tables 1–3.
The inverse relationship between nasopharyngeal col- onization and age may be related to the increased risk of respiratory infections in infants and young children since colonization increases significantly during periods of upper respiratory illness. This increase is especially prominent among children who are prone to recurrent otitis media.10,47
This can be explained by close contact between young children coupled with poorly developed immunity to these organisms.2
Adults with chronic respiratory diseases have higher carriage rates.7,45 A study carried out by Klingman et al.5 in patients with bronchiectasis showed a rate of colonization by M. catarrhalis of 43%, similar to that found in children. Each patient was colonized with one to four different strains for an average time of 2.3 months. In contrast, Zalacain et al.45 showed M. catarrhalis colonization rates of 10.4% in patients with stable COPD.
Higher isolation rates in soldiers may reflect high rates of transmission of respiratory organisms in the military setting, resembling the high rate of cross-infection among children in day-care centres.21 However, Christenson et al.20 demon- strated lower colonization rates in child day-care personnel.
Race. Factors other than race may contribute to differences in colonization: living conditions, socio-economic status, access to medical care, season, microbiological methods and criteria. Studies in various races have been undertaken to assess differences in colonization but this factor remains unresolved.40,46,83
Gender. In the majority of studies, rates of colonization did not differ significantly between boys and girls.13,28,41,47,84
Carriage of S. pneumoniae with decreased antibiotic susceptibility is unaccountably more common in females.15,39,76
Socio-economic status. Low socio-economic status has also been identified as a risk for colonization and carriage of respiratory pathogens.46,83,85 Probably cramped and poor housing associated with lower access to health care are the factors. H. influenzae and S. pneumoniae colonization rates of 65.4% and 55.7%, respectively, have been reported in children living in an area of 1.75 m2 per person, compared with 5.8% (H. influenzae) and 10.8% (S. pneumoniae) observed in children living in an area of 10.2 m2 per person.83 Coles et al.39 have reported a close relationship between low educational level of parents and higher nasopharyngeal colonization. Other studies have reported no relationship between carriage and this variable.13,40,75
Diet (breast-feeding versus bottle-feeding). Breast-feeding has been shown to protect against otitis media in several studies.10,86 However, it does not appear to have a substantial influence on nasopharyngeal colonization with respiratory pathogens.10,28,87 Duffy et al.88 have shown lower rates of colonization at 6 months of age in exclusively breast-fed children but not at 3 and 12 months of age.
Season. Respiratory pathogens can be recovered from the nasopharynx of healthy children and adults throughout the year.47,73 However, for reasons that remain unclear, the rate of colonization is seasonal, with an increase in midwinter.76 This is probably related to closer interpersonal contact (school year, indoor activities), lack of adequate ventilation, viral infections and use of antibiotics. In other studies, seasonal effects are insignificant or absent.47,73,84
Smoking history of parents. Although the effect of parental smoking on colonization is controversial, this habit has been identified as a factor associated with increased carriage of respiratory pathogens.10,39,83 An important implied factor is possibly the level of exposure. This can be influenced by a number of factors, for example, amount smoked in the pres- ence of the child, ventilation and so forth. In addition, infants may be exposed to passive smoke from other non-parental sources. Several mechanisms could explain the relationship between exposure to passive smoking and colonization by respiratory pathogens among children. Smoke may damage and inflame nasopharyngeal mucosa, increasing suscep- tibility to viral and bacterial colonization.
Exposure to other children. This has also been reported as a risk factor for carriage of respiratory pathogens.10,22,28 Day- care centres, orphanages and other crowded situations facilitate horizontal transfer of bacteria from one child to another.19,20,22,25,28,29,31,32,35,43,85
Family size also has been related to high colonization rates, presumably by increasing the potential exposure to infectious agents.19,28 In children who remain at home the older siblings are the main source of the nasopharyngeal microflora. The colonization rates with S. pneumoniae and/or H. influenzae and/or M. catarrhalis are higher among infants with siblings than in single children.19,28,30,39,40,43,85 Children probably acquire the bacteria in day care and introduce them at home.19
Other studies have reported no association between coloniza- tion and number of siblings.13
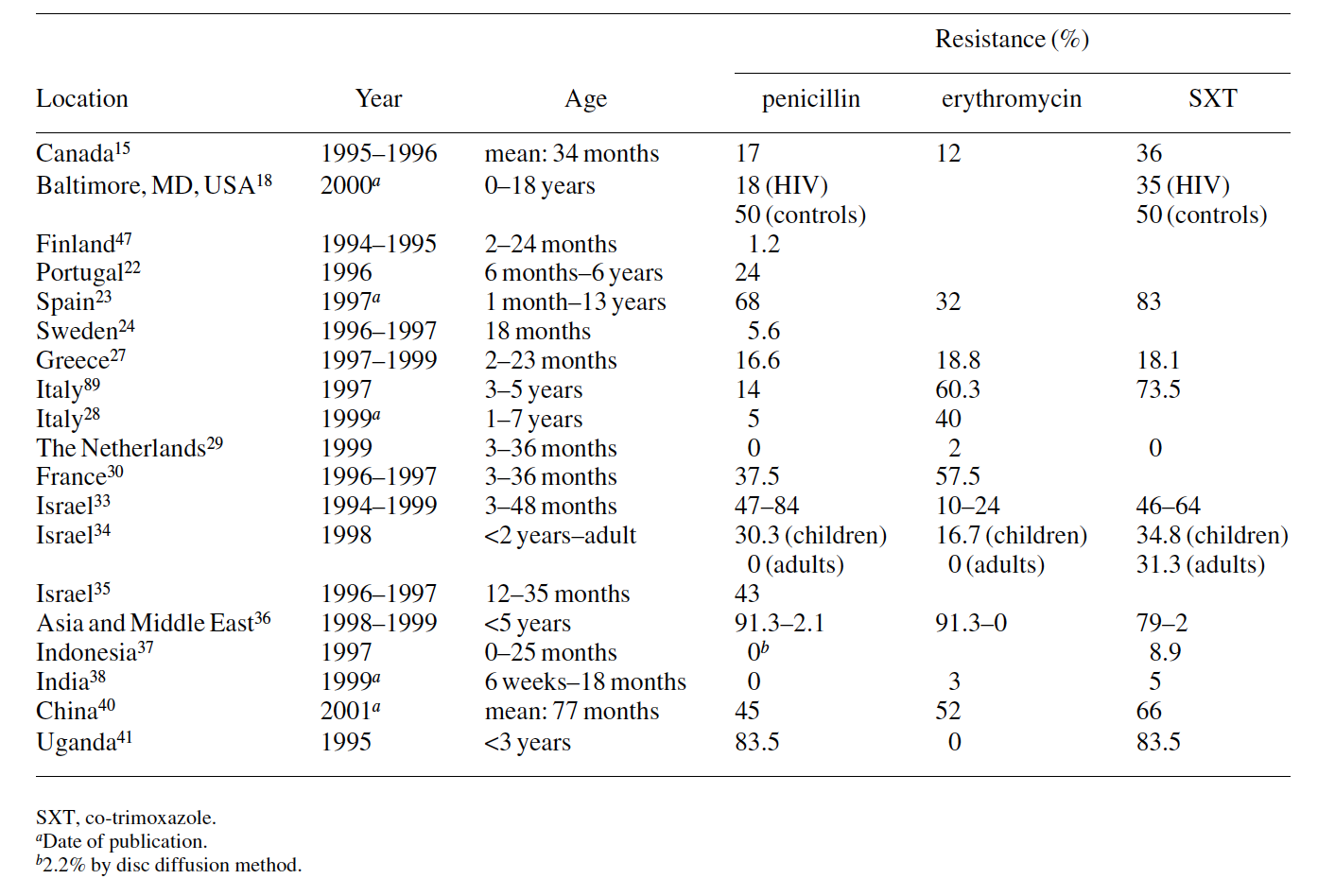
Many studies have demonstrated a strong association between day-care attendance and increased carriage of potential pathogens. Carriage is significantly higher in centres where ≥45 children attend than in centres with <45 children.2 On the other hand, the rates of colonization are higher following prolonged full-time attendance.28 Furthermore, these studies have shown frequent carriage of resistant strains. Day- care centres are efficient environments for the transmission of pathogens. In these centres, three conditions exist that favour the development and transmission of resistant bacteria: a large number of children, frequent close person-to-person contacts and intensive antimicrobial use, which seems to be particularly important in the selection of antibiotic-resistant strains. Intensive antimicrobial usage provides selective pres- sure that favours the appearance of resistant bacteria, whereas the day-care environment fosters the transmission of these strains. Thus, when children attending a day-care centre are exposed to antibiotics, the pool of susceptible organisms is replaced by a population of bacteria with decreased antibiotic susceptibility. Other day-care attendees can be colonized by resistant organisms even if they themselves are not being given antimicrobials.76 Children in day-care centres may constitute an important reservoir of resistant organisms and may contribute to the spread of resistant bacteria in the community. Many studies conducted in day-care centres have found high rates of nasopharyngeal carriage of resistant bacteria, albeit with local variations. In these institutions the isolation rates of pneumococci with reduced susceptibility to penicillin range between 1% and 95%,15,20,22–24,27,28,31,34–36,40,76,77,89 and the vast majority of these organisms belonged to a few serotypes, especially 6B, 14, 19 and 23F.22,23,27,34,35,47 An additional 2–60% of isolates showed resistance to one or more antimicrobial agents.15,20,22,27,76 Erythromycin resist- ance rates of between 16% and 67% have been reported in several parts of the world.22,23,26,28,40,89 Co-trimoxazole resist- ance rates of 14–87% have been reported.15,23,36,40,76,89 These ranges demonstrate wide variations in susceptibility from country-to-country, city-to-city and within segments of the population or even within day-care centres in a single city.22,35 The majority (90–100%) of the M. catarrhalis and 6–90% of
- influenzae isolated in these institutions are β-lactamase producers.22,28,30,43,77
Sleeping position. Harrison et al.84 have shown that prone sleeping has a marked effect on the nasopharyngeal flora and this could be related to the increased risk of sudden infant death syndrome in infants who sleep prone. In this sleeping position, secretions will pool in the upper airways leading to an increase in bacterial growth. The influence of sleeping position may be exacerbated by upper respiratory tract infection, which causes increased production of secretions and decreases the effectiveness of mucociliary clearance.
Upper respiratory tract infections. Many studies have shown that during periods of upper respiratory illness, colonization with potential respiratory pathogens increases significantly,47 but others failed to find such a connection.28 Viral infections induce impairment of the local host defences and may facilitate colonization of the respiratory tract by potential respiratory pathogenic bacteria. Likewise, viral infections are known to spread rapidly in day-care centres. Syrjanen et al.47 have shown than carriage of S. pneumoniae increased during respiratory infections (excluding AOM) from 13–43%, according to age group, to 45–56%.
Otitis media. A strong correlation between the naso- pharyngeal flora and middle ear infections has been found. In this way, nasopharyngeal colonization by H. influenzae, S. pneumoniae and/or M. catarrhalis in the first year of life is associated with an increased risk of recurrent otitis media com- pared with children who remain free of colonization.10,33,46
Another important reason for the high incidence of AOM among young children in particular seems to be very heavy colonization by potential pathogens in the nasopharynx in this age group. Potential pathogens may constitute >90% of the total bacterial count.90 In contrast, among healthy children viridans streptococci are predominant.91 Data from Syrjanen et al.47 show that S. pneumoniae carriage is lower during health (13–43%) than during AOM episodes, in particular during pneumococcal AOM (97–100%). Also, it has been shown that otitis-prone children tend to be colonized more often than non-otitis-prone children.74 On the other hand, nasopharyngeal colonization with resistant bacteria is associ- ated with an increased incidence of unresolved otitis media.
Sickle cell disease. Early studies found that patients with sickle cell disease had lower rates of nasopharyngeal carriage of S. pneumoniae than those in age-matched controls. More recently, the use of antibiotic prophylaxis reduced coloniza- tion rates but increased the number of resistant colonizing strains.16
HIV infection. Children with HIV infection are at higher risk for pneumococcal infection, having 12 times the risk of invasive disease of other children. This may be related to decreased mucosal immunity (IgA), which allows persistent colonization, or to decreased immunological responsiveness.2
For this reason, it is reasonable to assume greater carriage of pathogens in HIV-infected children. However, the few avail- able data contradict this hypothesis. Rusen et al.92 in Kenya, Leibovitz et al.93 in Romania and Polack et al.18 in the United States have demonstrated no differences in carriage between children with HIV infection and control groups. In contrast to these paediatric studies, studies of colonization in adults are conflicting.92–95
Therefore, in HIV-infected children, their immunocompromised state may not be an important predisposing condition for colonization. Antimicrobial prophylaxis appears to play a major role in controlling the pattern of colonization and antimicrobial resistance in strains from HIV-infected children.
Allergic constitution. In young children, allergy has been described as a risk factor for otitis media.96 Alternatively, allergic responses may result in impaired mucociliary activity, which may permit increased bacterial colonization of the nasopharynx. In a recent study, allergy was found to be associated with pneumococcal carriage in both adults and children.34 Also, multiple genetic types of H. influenzae can be detected in the upper respiratory tract of children with a history of allergy.75
Table 5. Changes in carriage of penicillin-intermediate or resistant S.pneumoniae after antimicrobial chemotherapy
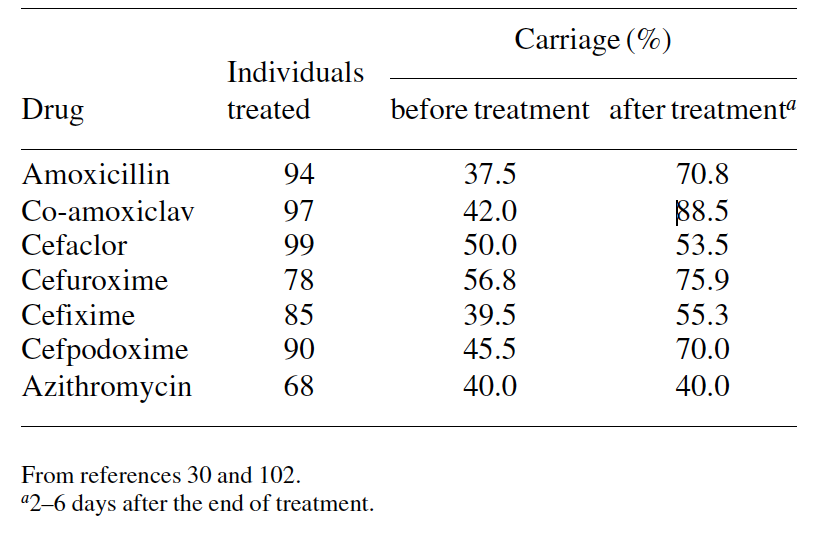
Antibiotic therapy. Antimicrobial treatment interferes with isolation rates of potential pathogens; lower isolation rates have been observed during and soon after antibiotic treatment, compared with pre-treatment rates. However, antimicrobial agents do not eradicate bacteria from the nasopharynx and the reduction in carriage is only temporary. After treatment, a rapid replacement of strains occurs with either over- growth of more resistant strains that were masked by other organisms or by newly acquired resistant strains.22,30,77,78,97–99
Thus, antibiotics induce several changes in nasopharyngeal flora: (i) suppression of susceptible bacteria, thereby creating an ecological niche for more resistant organisms; (ii) an increase in the number of resistant bacteria; (iii) an increase in the prevalence of antibiotic-resistant viridans streptococci, which constitute a reservoir of resistance genes that can be transferred to other bacterial species, such as S. pneumoniae; and (iv) they disturb the balance between pathogenic bacteria and α-haemolytic streptococci as a result of differential antibiotic susceptibility.2,100,101
However, the effects of antibiotics appear to differ accord- ing to the drug prescribed and the bacterial species. One may expect that drugs that have most antibacterial activity and greater penetration of the nasopharynx will achieve better eradication than those with less favourable pharmacokinetic/ pharmacodynamic properties. Antibiotics appear to be more active against M. catarrhalis and S. pneumoniae than against H. influenzae. Several studies demonstrate that H. influenzae carriage is only slightly affected by drug administration, including third-generation cephalosporins and co-amoxi- clav.77,78,98 In contrast, agents active against S. pneumoniae induced a drastic fall in the carriage of penicillin-susceptible pneumococci, thereby increasing the proportion of penicillin- resistant strains after treatment (Table 5).22,30,98,102 Naso- pharyngeal carriage of M. catarrhalis is significantly decreased by the most active drugs against this species.30 However, data from several studies have shown that the percentage of resistant and penicillin-resistant strains.99,102 Leach et al.97 showed prolonged carriage of S. pneumoniae after a single dose of azithromycin for the treatment of trachoma. Rates of carriage immediately before treatment and 2–3 weeks, 2 months and 6 months after treatment were 68%, 29%, 78% and 87%, respectively. Also, they showed a more rapid appearance of macrolide-resistant strains in previously colonized children than in previously uncolonized children.
Abdel-Haq et al.17 demonstrated an association between co-trimoxazole prophylaxis and nasopharyngeal colonization with both co-trimoxazole- and penicillin-resistant pneumo- cocci. The carriage of co-trimoxazole-resistant pneumococci increased from 13% in the control group to 64% in the study prophylaxis group. Also, decreased susceptibility to penicil- lin increased from 7% to 82% and resistance to erythromycin increased from 0% in the control group to 36% in the prophyl- axis group.
Thus, geographical variations in antimicrobial suscep- tibility can be explained by the general consumption of anti- microbial agents. Recent findings at an orphanage in Paris probably represent the peak of antibiotic resistance with a rate of penicillin resistance that exceeded 94%.31
The absence of resistant pneumococcal strains in The Netherlands found by Bogaert et al.29 (only 2% of the isolates were resistant to erythromycin, and no resistance to penicillin, co-trimoxazole or tetracycline was detected) may be ex- plained by the restricted use of antibiotics compared with that in many other countries. In contrast, the high rates of resist- ance to penicillin (83.5%) and co-trimoxazole (83.5%) found by Joloba et al.41 reflect the frequent prescription of these agents in Uganda (Table 6).
In conclusion, antibiotic use is the primary risk factor for carriage of resistant nasopharyngeal bacteria, particularly where exposure to low levels continues for prolonged periods. Youth has also been considered a risk factor for colonization with resistant bacteria,103 but day-care attendance and frequent use of empirical antibiotics are significant risk fac- tors. Other factors associated with a higher risk of antibiotic- resistant bacterial carriage include presence of older sib- lings,81,102,104,105 hospitalization and nosocomial acquisition, travel,40 winter months and urban living.28,37
Table 6. Nasopharyngeal carriage of resistant S. pneumoniae
Immunization. Studies of the impact of immunization on colonization by H. influenzae have yielded mixed results. In Gambia PRP-T (polyribosylribitol phosphate conjugate to tetanus toxoid; Pasteur-Merieux, Lyon, France) Hib vaccine administered during a vaccine trial led to a 60% reduction in H. influenzae type b carriage among vaccinees.11 In Brazil, children who had received adequate vaccination with PRP-T were four-fold less likely to be H. influenzae carriers (1.2% versus 4.8%).12 In Navajo and White Mountain Apache children, PRP-OMP (outer membrane protein conjugate) vaccination reduced H. influenzae carriage to very low lev- els.14 However, in a rural population of Alaska, a recent survey has found carriage rates of 2.2–13.2% despite high rates of vaccination.106 In contrast, in a nearby town in the same state, only 1.1% of the children were colonized with H. influenzae.107 These data suggest that the ability to eliminate H. influenzae type b carriage, transmission and disease may be affected by other factors such as vaccination cover- age, vaccination regimen, type of vaccine used and population characteristics.
Pneumococcal conjugate vaccines have been shown to reduce the acquisition and carriage of vaccine serotypes in the nasopharynx. A preliminary study carried out in Israel using a heptavalent pneumococcal conjugate vaccine showed a lower carriage of penicillin-resistant pneumococci in children re- ceiving this vaccine compared with children receiving non- conjugate vaccine.108 However, immunization can also place selective pressure on the nasopharyngeal flora, promoting the emergence of new pathogens. A shift towards non-vaccine serotypes has been observed among vaccinees in several stud- ies.109 In South Africa, the carriage of non-vaccine serotypes was increased from 24% in controls to 36% in the vaccines group. A significant increase was seen in the carriage of serotypes 7 and 15, important causes of invasive disease.109
Serotype replacement could be caused by two principal mechanisms: (i) vaccinated children who are exposed to both vaccine and non-vaccine serotypes are likely to be preferen- tially colonized by the non-vaccine strains because they are immunized against vaccine strains; and (ii) the reduction of vaccine strains could be due to the unmasking of serotypes carried in small numbers.110
The reduced carriage of vaccine serotypes in vaccinated children leads to an interruption of the transmission of these serotypes and would be expected to lead to herd immunity. In Israeli day-care centres, the carriage of vaccine serotypes was lower in the siblings of vaccinated children compared with the siblings of unvaccinated children.111 On the other hand, vaccination interrupts the transmission of antibiotic-resistant pneumococci and thus decreases the burden of antibiotic resistance in immunized children and in their contacts.111
Other factors. Other host factors associated with variation in the risk of nasopharyngeal colonization in children include use of pacifiers,75,84 hospitalization, conjunctivitis (H. influ- enzae)30 and xylitol consumption.112 Risk factors in adults are different from those found in children and include exposure to children at home or at the workplace, COPD, cystic fibrosis, smoking habit, alcohol intake, obesity, immunosuppression and acute sinusitis.21
References
- Rotimi, V. O. & Duerden, I. (1981). Development of the bacterial flora in normal neonates. Journal of Medical Microbiology 14, 51–62.
- Ghaffaar, F., Friedland, I. R. & McCracken, G. , Jr (1999). Dynamics of nasopharyngeal colonization by Streptococcus pneumoniae. Pediatric Infectious Disease Journal 18, 638–46.
- Faden, H., Duffy, L., Williams, , Krystofik, D. A. & Wolf, J. (1995). Epidemiology of nasopharyngeal colonization with non- typeable Haemophilus influenzae in the first two years of life. Journal of Infectious Diseases 172, 132–5.
- Faden, H., Harabuchi, & Hong, J. J. (1994). Epidemiology of Moraxella catarrhalis in children during the first two years of life: relationship to otitis media. Journal of Infectious Diseases 169, 1312–7.
- Klingman, K. L., Pye, , Murphy, T. F. & Hill, S. L. (1995). Dynamics of respiratory tract colonization by Moraxella (Branham- ella) catarrhalis in bronchiectasis. American Journal of Respiratory Critical Care Medicine 152, 1072–8.
- Samuelson, A., Freijd, A., Jonasson, & Lindberg, A. A. (1995). Turnover of nonencapsulated Haemophilus influenzae in the nasopharynges of otitis-prone children. Journal of Clinical Microbiology 33, 2027–31.
- Karalus, R. & Campagnari, A. (2000). Moraxella catarrhalis: a review of an important human mucosal pathoge Microbes and Infection 2, 547–59.
- Faden, H., Stanievich, J., Brodsky, & Berstein, J. M. (1990). Changes in nasopharyngeal flora during otitis media of childhood. Pediatric Infectious Disease Journal 9, 623–6.
- Faden, H., Duffy, L., Williams, D. , Krystofik, D. & Wolf, J. (1996). Epidemiology of nasopharyngeal colonization with non- typeable Haemophilus influenzae in the first two years of life. Acta Otolaryngologica Supplementum 523, 128–9.
- Faden, H., Duffy, L., Wasielewski, R., Wolf, J., Krystofik, D. A., Tung, Y. et al. (1997). Relationship between nasopharyngeal colonization and the development of otitis media in children. Journal of Infectious Diseases 175, 1440–5.
- Adegbola, R. A., Mulholland, E. K., Secka, O., Jaffar, S. & Greenwood, B. M. (1998). Vaccination with a Haemophilus influenzae type b conjugate vaccine reduces oropharyngeal car- riage of Haemophilus influenzae type b among Gambian children. Journal of Infectious Diseases 177, 1758–61.
- Forleo-Neto, E., de Oliveira, C. F., Maluf, E. M., Bataglin, C., Araujo, J. M., Kunz, L. F., Jr et al. (1999). Decreased point prevalence of Haemophilus influenzae type b (Hib) oropharyngeal colonization by mass immunization of Brazilian children less than 5 years old with Hib polyribosylribitol phosphate polysaccharide– tetanus toxoid conjugate vaccine in combination with diphtheria– tetanus toxoids–pertussis vaccine. Journal of Infectious Diseases 180, 1153–8.
- Fontanals, D., Bou, R., Pons, I., Sanfeliu, I., Domínguez, A., Pineda, V. et al. (2000).Prevalence of Haemophilus influenzae carriers in the Catalan preschool population. European Journal of Clinical Microbiology and Infectious Diseases 19, 301–4.
- Millar, E. V., O′Brien, K. L., Levine, O. S., Kvamme, S., Reid, R. & Santosham, M. (2000). Toward elimination of Haemophilus influenzae type b carriage and disease among high-risk American Indian children. American Journal of Public Health 90, 1550–4.
- Kellner, J. D., Ford-Jones, L., Matlow, A., Low, D. E., Corey, M., Watson, H. et al. (1996). Nasopharyngeal carriage of Streptococcus pneumoniae in children in day care centers. In Program and Abstracts of the Thirty-sixth Interscience Conference on Antimicro- bial Agents and Chemotherapy, New Orleans, LA, USA. Abstract K17, p. 252. American Society for Microbiology, Washington, DC, USA.
- Steele, R. W., Warrier, R., Unkel, P. J., Foch, B. J., Howes, R. F., Shah, S. et al. (1996). Colonization with antibiotic-resistant Streptococcus pneumoniae in children with sickle cell disease. Journal of Pediatrics 128, 531–5.
- Abdel-Haq, N., Abuhammour, W., Asmar, B., Thomas, R., Dabbagh, S. & Gonzalez, R. (1999). Nasopharyngeal colonization with Streptococcus pneumoniae in children receiving trimethoprim– sulfamethoxazole prophylaxis. Pediatric Infectious Disease Journal 18, 647–9.
- Polack, F. P., Flayhart, D. C., Zahural, M. L., Dick, J. D. & Willoughby, R. E. (2000). Colonization by Streptococcus pneumo- niae in human immunodeficiency virus-infected children. Pediatric Infectious Disease Journal 19, 608–12.
- Vives, M., García, M. E., Saenz, P., Mora, M. A., Mata, L., Sabharwal, H. et al. (1997). Nasopharyngeal colonization in Costa Rican children during the first year of life. Pediatric Infectious Disease Journal 16, 852–8.
- Christenson, B., Sylvan, S. P. & Noreen, B. (1997). Carriage of multiresistant Streptococcus pneumoniae among children attending day-care centers in the Stockholm area. Scandinavian Journal of Infectious Diseases 29, 555–8.
- Jousimies-Somer, H. R., Savolainen, S. & Ylikoski, J. S. (1989). Comparison of the nasal bacterial floras in two groups of healthy subjects and in patients with acute maxillary sinusitis. Journal of Clinical Microbiology 27, 2736–43.
- De Lencastr H., Kristinsson, K. G., Brito-Avo, A., Sanches, I. S., Sa-Leao, R., Saldaña, J. et al. (1999). Carriage of respiratory tract pathogens and molecular epidemiology of Streptococcus pneumoniae colonization in healthy children attending day care centers in Lisbon, Portugal. Microbial Drug Resistance 5, 19–29.
- García-de-Lomas, J., Gimeno, , Millás, E., Bermejo, M., Lázaro, M. A., Navarro, D. et al. (1997). Antimicrobial susceptibility of Streptococcus pneumoniae isolated from pediatric carriers in Spain. European Journal of Clinical Microbiology and Infectious Diseases 16, 11–3.
- Borres, M. , Alestig, K., Krantz, I., Larsson, P., Norvenius, G. & Stenqvist, K. (2000). Carriage of penicillin-susceptible and non- susceptible pneumococci in healthy young children in Göteborg, Sweden. Journal of Infection 40, 141–4.
- Gunnarsson, R. K., Holm, S. E. & Soderstrom, M. (1998). The prevalence of potential pathogenic bacteria in nasopharyngeal samples from healthy children and adults. Scandinavian Journal of Primary Health Care 16, 13–
- Syrogiannopoulos, G. A., Grivea, I. , Davies, T. A., Katopodis, G. D., Appelbaum, P. C. & Beratis, N. G. (2000). Antimicrobial use and colonization with erythromycin-resistant Streptococcus pneumo- niae in Greece during the first 2 years of life. Clinical Infectious Diseases 31, 887–93
- Syrogiannopoulos, G. A., Grivea, I. , Katopodis, G. D., Geslin, P., Jacobs, M. R. & Beratis, N. G. (2000). Carriage of antibiotic- resistant Streptococcus pneumoniae in Greek infants and toddlers. European Journal of Clinical Microbiology and Infectious Diseases 19, 288–93.
- Principi, , Marchisio, P., Schito, G. C. & Mannelli, S. (1999). Risk factors for carriage of respiratory pathogens in the naso- pharynx of healthy children. Ascanius Project Collaborative Group. Pediatric Infectious Disease Journal 18, 517–23.
- Bogaert, , Engelen, M. N., Timmers-Reker, A. J. M., Elzenaar, K. P., Peerbooms, P. G. H., Coutinho, R. A. et al. (2001). Pneumococcal carriage in children in the Netherlands. A molecular epidemiological study. Journal of Clinical Microbiology 39, 3316–20.
- Varon, E., Levy, , La Rocque, F., Boucherat, M., Deforche, D., Podglajen, I. et al. (2000). Impact of antimicrobial therapy on nasopharyngeal carriage of Streptococcus pneumoniae, Haemo- philus influenzae, and Branhamella catarrhalis in children with respiratory tract infections. Clinical Infectious Diseases 31, 477–81.
- Raymond, J., Le Thomas, , Moulin, F., Commeau, A., Gendrel, D. & Berche, P. (2000). Sequential colonization by Streptococcus pneumoniae of healthy children living in an orphanage. Journal of Infectious Diseases 181, 1983–8.
- Stratchounsky, S., Kretchikova, O. I., Kozlov, R. S., Reshedko, G. K., Stetsiouk, O. U., Tarasova, G. D. et al. (2000). Antimicrobial resistance of Streptococcus pneumoniae isolated from healthy children in day care centers: results of a multicenter study in Russia. Pediatric Infectious Disease Journal 19, 196–200.
- Eldan, M., Leibovitz, , Piglanski, L., Raiz, S., Press, J., Yagupsky, P. et al. (2000). Predictive value of pneumococcal nasopharyngeal cultures for the assessment of nonresponsive acute otitis media in children. Pediatric Infectious Disease Journal 19, 298–303.
- Borer, , Meirson, H., Peled, N., Porat, N., Dagan, R., Fraser, D. et al. (2001). Antibiotic-resistant pneumococci carried by young children do not appear to disseminate to adult members of a closed community. Clinical Infectious Diseases 33, 436–44.
- Givon-Lavi, N., Dagan, R., Fraser, D., Yagupsky, P. & Porat, N. (1999). Marked differences in pneumococcal carriage and resist- ance patterns between day care centers located within a small area. Clinical Infectious Diseases 29, 1274–80.
- Lee, N. Y., Song, J. H., Kim, S., Peck, K. R., Ahn, K. M., Lee, S. et al. (2001). Carriage of antibiotic-resistant pneumococci among asian children: a multinational surveillance by the Asian Network for Surveillance of Resistant Pathogens (ANSORP). Clinical Infectious Diseases 32, 1463–9.
- Soewignjo, S., Gessner, B. D., Sutanto, A., Steinhoff, M., Prijanto, M., Nelson, C. et al. (2001). Streptococcus pneumoniae nasopharyngeal carriage prevalence, serotype distribution, and resistance patterns among children on Lombok Island, Indonesia. Clinical Infectious Diseases 32, 1039–43.
- Jebaraj, R., Cherian, T., Raghupathy, P., Brahmadathan, K. N., Lalitha, M. K., Thomas, K. et al. (1999). Nasopharyngeal coloniza- tion of infants in southern India with Streptococcus pneumoniae. Epidemiology and Infection 123, 383–8.
- Coles, C. L., Kanungo, R., Rahmathullah, L., Thulasiraj, R. D., Katz, J., Santosham, M. et al. (2001). Pneumococcal nasopharyn- geal colonization in young South Indian infants. Pediatric Infectious Disease Journal 20, 289–95.
- Boost, M. V., O’Donoghue, M. M. & Dooley, J. S. (2001). Prevalence of carriage of antimicrobial resistant strains of Strepto- coccus pneumoniae in primary school children in Hong Kong. Epidemiology and Infection 127, 49–55.
- Joloba, M. L., Bajaksouzian, S., Palavecino, E., Whalen, C. & Jacobs, M. R. (2001). High prevalence of carriage of antibiotic- resistant Streptococcus pneumoniae in children in Kampala, Uganda. International Journal of Antimicrobial Agents 17, 395–400.
- Adegbola, R. A., Obaro, S. K., Biney, E. & Greenwood, B. M. (2001). Evaluation of Binax now Streptococcus pneumoniae urinary antigen test in children in a community with a high carriage rate of pneumococcus. Pediatric Infectious Disease Journal 20, 718–9.
- Raymond, J., Armand-Lefevre, L., Moulin, F., Dabernat, H., Commeau, A., Gendrel, D. et al. (2001). Nasopharyngeal coloniza- tion by Haemophilus influenzae in children living in an orphanage. Pediatric Infectious Disease Journal 20, 779–84.
- Berner, R., Schumacher, R. F., Brandis, M. & Forster, J. (1996). Colonization and infection with Moraxella catarrhalis in childhood. European Journal of Clinical Microbiology and Infectious Diseases 15, 506–9.
- Zalacain, R., Sobradillo, V., Amilibia, J., Barron, J., Achotegui, V., Pijoan, J. I. et al. (1999). Predisposing factors to bacterial colonization in chronic obstructive pulmonary disease. European Respiratory Journal 13, 232–5.
- Leach, A. J., Boswell, J. B., Asche, V., Nienhuys, T. G. & Mathews, J. D. (1994). Bacterial colonization of the nasopharynx predicts very early onset and persistence of otitis media in Australian Aboriginal infants. Pediatric Infectious Disease Journal 13, 983–9.
- Syrjanen, R. K., Kilpi, T. M., Kaijalainen, T. H., Herva, E. E. & Takala, A. K. (2001). Nasopharyngeal carriage of Streptococcus pneumoniae in Finnish children younger than 2 years old. Journal of Infectious Diseases 184, 451–9.
- Harabuchi, , Faden, H., Yamanaka, N., Duffy, L., Wolf, J. & Krystofik, D. (1994). Nasopharyngeal colonization with nontypeable Haemophilus influenzae and recurrent otitis media. Journal of Infectious Diseases 170, 862–6.
- Rapola, S., Salo, , Kiiski, P., Leinonen, M. & Takala, A. K. (1997). Comparison of four different sampling methods for detecting pharyngeal carriage of Streptococcus pneumoniae and Haemo- philus influenzae in children. Journal of Clinical Microbiology 35, 1077–9.
- Musher, D. M. (2000). Streptococcus pneumoniae. In Principles and Practice of Infectious Diseases, 5th edn (Mandell, G. L., Bennett, E. & Dolin, R., Eds), pp. 2128–46. Churchill Livingstone, Philadelphia, PA, USA.
- 51. Austrian, R. (1986). Some aspects of the pneumococcal carrier stat Journal of Antimicrobial Chemotherapy 18, Suppl. A, 35–45.
- Krivan, H. C., Roberts, D. D. & Ginsburg, (1988). Many pulmonary pathogenic bacteria bind specifically to the carbohydrate sequence GalNac β 1-4 Gal found in some glycolipids. Proceedings of the National Academy of Sciences, USA 85, 6157–61.
- Barthelson, , Mobasseri, A., Zopf, D. & Simon, P. (1998). Adherence of Streptococcus pneumoniae to respiratory epithelial cells is inhibited by sialylated oligosaccharides. Infection and Immunity 66, 1439–44.
- Van der Flier, M., Chhun, N., Wizemann, T. M., Min, J., McCarthy, B. & Tuomanen, E. I. (1995). Adherence of Strepto- coccus pneumoniae to immobilized fibronectin. Infection and Immunity 63, 4317–22.
- Weiser, J., Austrian, , Sreenivasan, P. & Masure, H. R. (1994). Phase variation in pneumococcal opacity. Relationship between colonial morphology and nasopharyngeal colonization. Infection and Immunity 62, 2582–9.
- Kim, J. O. & Weiser, N. (1998). Association of intrastrain phase variation in quantity of capsular polysaccharide and teichoic acid with the virulence of Streptococcus pneumoniae. Journal of Infectious Diseases 177, 368–77.
- Weiser, N., Markiewicz, Z., Tuomanen, E. & Wani, J. H. (1996). Relationship between phase variation in colony morpho- logy, intrastrain variation in cell wall physiology, and naso- pharyngeal colonization by Streptococcus pneumoniae. Infection and Immunity 64, 2240–5.
- Cundell, D. R., Gerard, N. , Gerard, C., Idanpaan-Heikkila, I. & Tuomanen, E. I. (1995). Streptococcus pneumoniae anchor to activated human cells by the receptor for platelet-activating factor. Nature 377, 435–8.
- Cundell, D., Weiser, J., Shen, J., Young, A. & Tuomanen, E. (1995). Relationship between colonial morphology and adherence of Streptococcus pneumoniae. Infection and Immunity 63, 757–61.
- Reddy, M. S., Berstein, J. M., Murphy, T. & Faden, H. (1996). Binding between outer membrane proteins of nontypeable Haemo- philus influenzae and human nasopharyngeal mucin. Infection and Immunity 64, 1477–9.
- St Geme, J. W., III (2001). The pathogenesis of nontypeable Haemophilus influenzae otitis media. Vaccine 19, Suppl., S41–50.
- St Geme, W., III (1994). The HMW1 adhesin of nontypeable Haemophilus influenzae recognizes sialylated glycoprotein recep- tors on cultured human epithelial cells. Infection and Immunity 62, 3881–9.
- McMichael, J. C., Fiske, M. J., Fredenburg, R. A., Chakravatti, D. N., Vandermeid, K. R., Barniak, V. et al. (1998). Isolation and characterization of two proteins from Moraxella catarrhalis that bear a common epitope. Infection and Immunity 66, 4374–81.
- Aebi, C., Lafontaine, E. R., Cope, L. D., Latimer, J. L., Lumbley, S. L., McCracken, G. H., Jr et al. (1998). Phenotypic effect of isogenic uspA1 and uspA2 mutations on Moraxella catarrhalis O35E. Infection and Immunity 66, 3113–9.
- McMichael, J. C. (2000). Vaccines for Moraxella catarrhalis. Vaccine 19, Suppl., S101–7.
- Faden, H. (2001). The microbiologic and immunologic basis for recurrent otitis media in children. European Journal of Pediatrics 160, 407–13.
- Simell, B., Korkeila, M., Pursiaenen, H., Kilpi, T. M. & Kayhty, M. (2001). Pneumococcal carriage and otitis media induce salivary antibodies to pneumococcal surface adhesin A, pneumolysin, and pneumococcal surface protein A in children. Journal of Infectious Diseases 183, 887–96.
- Rapola, S., Janntti, V., Haikala, R., Syrjanen, R., Carlone, G. M., Sampson, J. S. et al. (2000). Natural development of antibodies to pneumococcal surface protein A, pneumococcal surface adhesin A, and pneumolysin in relation to pneumococcal carriage and acute otitis media. Journal of Infectious Diseases 182, 1146–52.
- Samukawa, T., Yamanaka, N., Hollingshead, S., Klingman, K. & Faden, H. (2000). Immune responses to specific antigens of Streptococcus pneumoniae and Moraxella catarrhalis in the respiratory tract. Infection and Immunity 68, 1569–73.
- Soininen, A., Pursiainen, H., Kilpi, T. & Käyhty, H. (2001). Natural development of antibodies to pneumococcal capsular polysaccharides depends on the serotype: association with pneumococcal carriage and acute otitis media in young children. Journal of Infectious Diseases 184, 569–76.
- Tano, K., Grahn-Hakansson, E., Holm, S. E. & Hellstrom, S. (2000). Inhibition of OM pathogens by α-hemolytic streptococci from healthy children, children with SOM and children with rAOM. Inter- national Journal of Pediatric Otorhinolaryngology 56, 185–90.
- Gray, B. M. & Dillon, H. C., Jr (1988). Epidemiological studies of Streptococcus pneumoniae in infants: antibody to types 3, 6, 14 and 23 in the first two years of life. Journal of Infectious Diseases 158, 948–55.
- Marchissio, P., Gironi, S., Esposito, S., Schito, G. C., Mannelli, S., Principi, N. et al. (2001). Seasonal variations in nasopharyngeal carriage of respiratory pathogens in healthy Italian children attend- ing day-care centres or schools. Journal of Medical Microbiology 50, 1095–9.
- Dhooge, I., Vaneechoutte, M., Claeys, G., Verschraegen, G. & Van Cauwenberge, P. (2000). Turnover of Haemophilus influenzae isolates in otitis-prone children. International Journal of Pediatric Otorhinolaryngology 54, 7–12.
- Sauver, J. S., Marrs, C. F., Foxman, B., Somsel, P., Madera, R. & Gilsdorf, J. R. (2000). Risk factors for otitis media and carriage of multiple strains of Haemophilus influenzae and Streptococcus pneumoniae. Emerging Infectious Diseases 6, 622–30.
- Yagupsky, P., Porat, N., Fraser, D., Prajgrod, F., Merires, M., McGee, L. et al. (1998). Acquisition, carriage, and transmission of pneumococci with decreased antibiotic susceptibility in young children attending a day care facility in southern Israel. Journal of Infectious Diseases 177, 1003–12.
- Dabernat, , Geslin, P., Megraud, F., Begue, P., Boulesteix, J., Dubreuil, C. et al. (1998). Effects of cefixime or co-amoxiclav treat- ment on nasopharyngeal carriage of Streptococcus pneumoniae and Haemophilus influenzae in children with acute otitis media. Journal of Antimicrobial Chemotherapy 41, 253–8.
- Cohen, , Navel, M., Grumberg, J., Boucherat, M., Geslin, P., Derriennic, M. et al. (1999). One dose ceftriaxone vs ten days of amoxicillin/clavulanate therapy for acute otitis media: clinical efficacy and change in nasopharyngeal flora. Pediatric Infectious Disease Journal 18, 403–9.
- Spinola, S. M., Peacock, , Denny, F. W., Smith, D. L. & Cannon, J. G. (1986). Epidemiology of colonization by nontypeable Haemophilus influenzae in children: a longitudinal study. Journal of Infectious Diseases 154, 100–9.
- Ekdahl, K., Ahlinder, I., Hansson, H. , Melander, E., Molstad, S., Soderstrom, M. et al. (1997). Duration of nasopharyngeal carriage of penicillin-resistant Streptococcus pneumoniae: experi- ences from the South Swedish Pneumococcal Intervention Project. Clinical Infectious Diseases 25, 1113–7.
- Leiberman, A., Dagan, R., Leibovitz, E., Yagupsky, & Fliss, D. M. (1999). The bacteriology of the nasopharynx in childhood. International Journal of Pediatric Otorhinolaryngology 49, Suppl. 1, S151–3.
- Linsk, R. L., Hussain, R. S. & Ozor, C. E. (2000). Naso- pharyngeal colonization in healthy young adults. In Program and Abstracts of the Fortieth Interscience Conference on Antimicrobial Agents and Chemotherapy, Toronto, Canada, 2000. Abstract 1804, 112. American Society for Microbiology, Washington, DC, USA.
- Sung, R. T., Ling, J. M., Fung, S. M., Oppenheimer, S. J., Crook, D. W., Lau, J. T. et al. (1995). Carriage of Haemophilus influenzae and Streptococcus pneumoniae in healtly Chinese and Vietnamese children in Hong Kong. Acta Paediatrica 84, 1262–7.
- Harrison, L. M., Morris, J. , Telford, D. R., Brown, S. M. & Jones, K. (1999). The nasopharyngeal bacterial flora in infancy: effects of age, gender, season, viral upper respiratory tract infection and sleeping position. FEMS Immunology and Medical Microbiology 25, 19–28.
- Anianson, , Alm, B., Andersson, B., Larsson, P., Nylen, O., Peterson, H. et al. (1992). Nasopharyngeal colonization during the first year of life. Journal of Infectious Diseases 165, Suppl. 1, S38–42.
- Anianson, G., Alm, , Andersson, B., Hakansson, A., Larsson,. P., Nylen, O. et al. (1994). A prospective cohort study on breast- feeding and otitis media in Swedish infants. Pediatric Infectious Dis- ease Journal 13, 183–8.
- Kaleida, P. , Nativio, D. G., Chao, H. P. & Cowden, S. N. (1993). Prevalence of bacterial respiratory pathogens in the nasopharynx in breast-fed versus formula-fed infants. Journal of Clinical Microbiology 31, 2674–8.
- Duffy, C., Faden, H., Wasielewski, R., Wolf, J. & Krystofik, D. (1997). Exclusive breastfeeding protects against bacterial coloniza- tion and day care exposure to otitis media. Pediatrics 100, E7.
- Ronchetti, M. P., Guglielmi, F., Latini, L., Merolla, R., Lorusso, G., Bajaksouzian, S. et al. (1999). Resistance patterns of Streptococcus pneumoniae from children in Central Italy. European Journal of Clinical Microbiology and Infectious Diseases 18, 376–9.
- Stenfors, E. & Raisanen, S. (1990). Occurrence of middle ear pathogens in the nasopharynx of young individuals. A quantitat- ive study in four age groups. Acta Otolaryngologica 109, 142–8.
- Long, S., Henretig, F. M., Teter, M. J. & McGowan, K. L. (1983). Nasopharyngeal flora and acute otitis media. Infection and Immunity 41, 987–91.
- Rusen, I. , Fraser-Roberts, L., Slaney, L., Ombette, J., Lovgren, M., Datta, P. et al. (1997). Nasopharyngeal pneumococcal colonization among Kenyan children: antibiotic resistance, strain types and associations with human immunodeficiency virus type 1 infection. Pediatric Infectious Disease Journal 16, 656–62.
- Leibovitz, E., Dragomir, C., Sfartz, S., Porat, , Yagupsky, P., Jica, S. et al. (1999). Nasopharyngeal carriage of multidrug- resistant Streptococcus pneumoniae in institutionalized HIV-infected and HIV-negative children in northeastern Romania. International Journal of Infectious Diseases 3, 211–5.
- Janoff, E. , O’Brien, J., Thompson, P., Ehret, J., Meiklejohn, G., Duvall, G. et al. (1993). Streptococcus pneumoniae coloniza- tion, bacteremia, and immune response among persons with human immunodeficiency virus infection. Journal of Infectious Diseases 167, 49–56.
- Rodriguez-Barradas, M. , Tharapel, R. A., Groover, J. E., Giron, K. P., Lacke, C. E., Houston, E. D. et al. (1997). Colonization by Streptococcus pneumoniae among human immunodeficiency virus-infected adults: prevalence of antibiotic resistance, impact of immunization, and characterization by polymerase chain reaction with BOX primers of isolates from persistent S. pneumoniae carriers. Journal of Infectious Diseases 175, 590–7.
- Kvaerner, K. J., Tambs, , Harris, J. R., Mair, I. W. & Magnus, P. (1996). Otitis media: relationship to tonsillitis, sinusitis, and atopic diseases. International Journal of Pediatric Otorhinolaryngology 35, 127–41.
- Leach, J., Shelby-James, T. M., Mayo, M., Gratten, M., Laming, A. C., Currie, B. J. et al. (1997). A prospective study of the impact of community-based azithromycin treatment of trachoma on carriage of Streptococcus pneumoniae. Clinical Infectious Diseases 24, 356–62.
- Cohen, , Bingen, E., Varon, E., de La Rocque, F., Brahimi, N., Levy, C. et al. (1997). Change in nasopharyngeal carriage of Streptococcus pneumoniae resulting from antibiotic therapy for acute otitis media in children. Pediatric Infectious Disease Journal 16, 555–60.
- Dagan, , Leibovitz, E., Greenberg, D., Yagupsky, P., Fliss, D. M. & Leibergman, A. (1998). Dynamics of pneumococcal naso- pharyngeal colonization during the first days of antibiotic treatment in pediatric patients. Pediatric Infectious Disease Journal 17, 880–5.
- Ghaffaar, , Friedland, I. R., Katz, K., Muniz, L. S., Smith, J. L., Davis, P. et al. (1999). Increased carriage of resistant non-pneumococcal α-hemolytic streptococci after antibiotic therapy. Journal of Pediatrics 135, 618–23.
- Ioannidou, S., Tassios, P. T., Kotsovili-Tseleni, , Foustoukou, M., Legakis, N. J. & Vatapoulos, A. (2001). Antibiotic resistance rates and macrolide resistance phenotypes of viridans group streptococci from the oropharynx of healthy Greek children. Inter- national Journal of Antimicrobial Agents 17, 195–201.
- Ghaffaar, F., Muniz, L. S., Katz, K., Reynolds, J., Smith, J. L., Davis, P. et al. (2000). Effects of amoxicillin/clavulanate or azithro- mycin on nasopharyngeal carriage of Streptococcus pneumoniae and Haemophilus influenzae in children with acute otitis media. Clinical Infectious Diseases 31, 875–80.
- Melander, E., Mölstad, S., Persson, K., Hansson, H. B., Söder- ström, M. & Ekdahl, K. (1998). Previous antibiotic consumption and other risk factors for carriage of penicillin-resistant Streptococcus pneumoniaae in children. European Journal of Clinical Microbiology and Infectious Diseases 17, 834–8.
- Ciftci, E., Dogru, U., Aysev, D., Ince, E., Guriz, H. & Aysev, U. D. (2001). Investigation of risk factors for penicillin-resistant Streptococcus pneumoniae carriage in Turkish children. Pediatrics International 43, 385–90.
- Samore, M. H., Magill, M. K., Alder, S. C., Severina, E., Morrison de Boer, L., Lyon, J. L. et al. (2001). High rates of multiple antibiotic resistance in Streptococcus pneumoniae from healthy children living in isolated rural communities: association with cephalosporin use and intrafamilial transmission. Pediatrics 108, 856–65.
- Galil, K., Singleton, R., Levine, O. S., Fitzgerald, M. A., Bulkow, L., Getty, M. et al. (1999). Reemergence of invasive Haemophilus influenzae type b disease in a well-vaccinated popula- tion in remote Alaska. Journal of Infectious Diseases 179, 101–6.
- Singleton, R., Bulkow, L. R., Levine, O. S., Hennessy, T., Butler, J. C. & Parkinson, A. J. (2000). Experience with prevention of invasive Haemophilus influenzae type b disease by vaccination in Alaska: the impact of persistent oropharyngeal carriage. Journal of Pediatrics 137, 313–20.
- Dagan, , Melamed, R., Muallem, M., Piglansky, L., Green- berg, D., Abramson, O. et al. (1996). Reduction of nasopharyngeal carriage of pneumococci during the second year of life by a hepta- valent conjugate pneumococcal vaccine. Journal of Infectious Diseases 174, 1271–8.
- Mbelle, N., Huebner, R. , Wasas, A. D., Kimura, A., Chang, I. & Klugman, K. P. (1999). Immunogenicity and impact on naso- pharyngeal carriage of nonavalent pneumococcal conjugate vaccine. Journal of Infectious Diseases, 180, 1171–6.
- Klugman, K. (2001). Efficacy of pneumococcal conjugate vaccines and their effect on carriage and antimicrobial resistance. Lancet Infectious Diseases 1, 85–91.
- Dagan, , Givon-Lavi, M., Porat, M., Sikuler-Cohen, M. & Fraser, D. (2000). Immunization of toddlers attending day care centers (DCCs) with a nine-valent conjugate pneumococcal vaccine (PncCRM9) reduces transmission of Streptococcus pneumoniae (Pnc) and antibiotic-resistant S. pneumoniae (R-Pnc) to their young siblings. In Program and Abstracts of the Fortieth Interscience Conference on Antimicrobial Agents and Chemotherapy, Toronto, Canada, 2000. Abstract 687, p. 244. American Society for Micro- biology, Washington, DC, USA.
- Uhari, M., Tapiainen, & Kontiokari, T. (2000). Xylitol as a non-vaccine prophylaxis for acute otitis media. Vaccine 19, Suppl. 1, S144–7.
José Ángel García-Rodríguez* and Mariá José Fresnadillo Martínez
Departamento de Microbiología, Hospital Universitario de Salamanca, Facultad de Medicina, Ps. S. Vicente 108,
37007 Salamanca, Spain